Companion Diagnostic Solutions
Our mission is to improve patients’ quality of life by expanding access to precision medicine through reliable Companion Diagnostics (CDx).
As a global companion diagnostics partner, we simplify regulatory complexity and accelerate CDx project development through fully customized solutions and transparent, end-to-end collaboration, so pharmaceutical teams can stay focused on what matters most: bringing targeted therapies to patients in an increasingly regulated market.
Companion diagnostics are an important element of certain precision medicine therapies. A CDx can provide both healthcare provider and patient, with the specific biomarker information to assess whether particular therapies can benefit the patient, and in some cases, whether a therapies might be harmful to the patient.
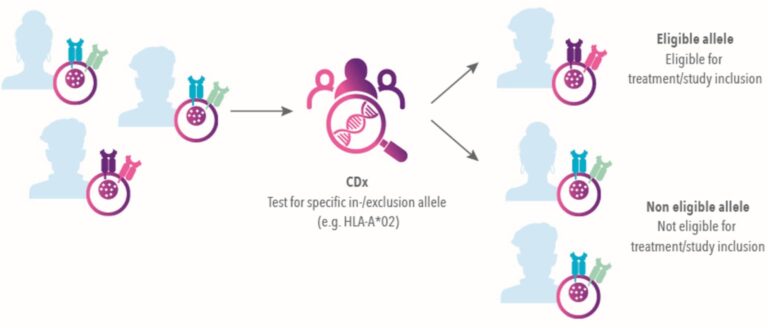
Companion diagnostics can also play a facilitating role in research, enabling faster, more tailored recruitment of eligible patients into clinical trials for precision therapies. In short, Companion Diagnostics allow:
Targeted patient identification
Identify patients most likely to benefit from your therapy
Risk stratification
Identify individuals at increased risk for serious adverse reactions
Therapy monitoring
Monitor treatment response for optimized safety and efficacy
Companion Diagnostics
Our Companion Diagnostics (CDx) services are designed to facilitate and support the pharmaceutical precision medicine revolution.
Our dedicated project team will work closely with you to ensure that every aspect of the project aligns with your specific requirements and the applicable legislation. Every project starts with a fully customized project design. From there on GenDx will guide and support you from idea, through the different stages of clinical implementation, to IVD. Our CDx services are distinctive for:
CE-marked molecular diagnostics under IVDR
NGS-based HLA typing reagents and analysis software
Rapid development timeline
From CE-marked legacy device to tailor-made clinical performance study assay in 6-9 months
Full-service CDx support
Facilitating bridging studies and interventional studies from idea to CDx
Proven regulatory experience
Europe and USA approvals, with global expansion capability
Broad assay development capabilities
Experience with developing diagnostic solutions for full gene analysis of complex, polymorphic genes, such as HLA, KIR, MICA/MICB and many more
Flexible, collaborative approach
Tailored solutions to meet your CDx needs
Strong global network
Supporting clinical studies & regulatory submissions
End-to-end expertise
Assay development including, manufacturing, supply chain and regulatory support
Reach out to us and learn how we can help you with your questions! We are always happy to set up a meeting and discuss possibilities and provide you with the input for your CDx strategy with GenDx.
Meet the Team
Our CDx team, part of the GenDx company, is comprised of experts with diverse backgrounds and extensive experience in molecular diagnostics, HLA Typing, regulatory affairs, and business development. With a shared commitment to innovation and excellence, we are dedicated to advancing personalized medicine and improving patient outcomes.
Partner with us today and embark on a journey towards precision healthcare tailored to the individual needs of each patient.
You ask, we answer
Our CDx team would like to help you!
What is a Companion Diagnostic device?
A companion diagnostic (CDx) test is defined as a medical device that provides information that is essential for the safe and effective use of a corresponding therapy. More specifically, a companion diagnostic is a diagnostic test that is clinically proven to accurately and reliably identify patients that are most likely to benefit from the associated therapy or at increased risk for serious adverse reactions from the associated therapy.
Why do you need to develop CDx for your therapeutic?
- Pharmacological clinical trials with patient selection as part of the inclusion/exclusion criteria and may need an investigational in vitro diagnostic (IVD) medical device for medical management decisions. A CDx can screen specific biomarkers and identify patients that are likely to respond to the therapy or patients that are at increased risk for adverse events.
- Including a CDx in your precision medicine strategy can increases the likelihood of a positive response and better clinical outcomes, as the CDx can match the right patients with the right treatment. The use of a CDx to pre-screen and select patients for clinical trials can lead to more efficient trials as the trial population is more targeted, possibly reducing the cost and shortening timelines.
How does GenDx support the development of a CDx from concept to clinical use?
- GenDx can facilitate the development of your requested assay from prototype concept to registered Companion Diagnostic IVD. Our team of Research & Development and Operational experts will support the development of a robust IVD and our Regulatory experts will facilitate an effective performance study application process for authorization of a clinical study to validate the clinical validity of the device.
- If applicable, your CDx can leverage technical validation of our currently CE marked devices for transplantation purposes, shortening development timelines. In alignment with your requirements, we will develop and design the clinical trial assay for use in interventional clinical trials or retrospective bridging studies. The IVD will be developed and validated, compliant with EU (IVDR) and/or US (FDA) regulations.
Which genes does GenDx offer a CDx for?
- GenDx can develop a CDx from Idea to IVD, for any gene of interest. Development trajectories and timelines differ when starting from a CE marked device (for different intended purposes), from a Research Use Only (RUO) device or from a prototype or concept. The technical validation of the CE marked and RUO solutions may be used for streamlined development.
- GenDx does already have CE marked devices for transplantation purposes targeting HLA-A, -B, -C, -DRB1, -DRB3/4/5, -DQB1, -DQA1, -DPB1 and DPA1. Additionally, GenDx offers RUO solutions for KIR, MICA and MICB gene analysis. Alternatively, we can use our expertise in molecular diagnostic solutions to design and develop your CDx from Idea to IVD. It follows that such a development from Idea to IVD will take longer than starting from a CE marked kit.
What kind of clinical studies can GenDx support?
- GenDx can support your therapy development in any stage, during the pre-clinical stage with RUO reagents and during the clinical stages, from first in human to the pivotal registrational trial. At the moment, that clinical device studies are required, GenDx can support interventional clinical studies (both prospective and retrospective) and clinical bridging studies.
- As IVD sponsor of the clinical study we facilitate performance study applications to the regulatory authorities and ethical bodies and we ensure clinical monitoring of the clinical testing site, through our strong network of partners.